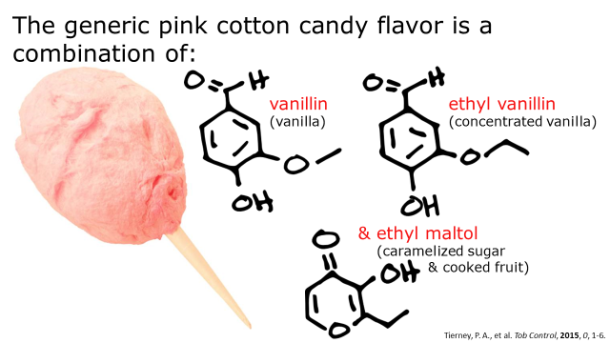
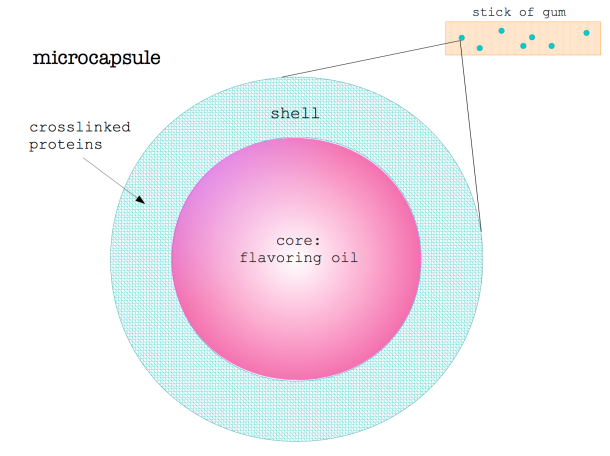
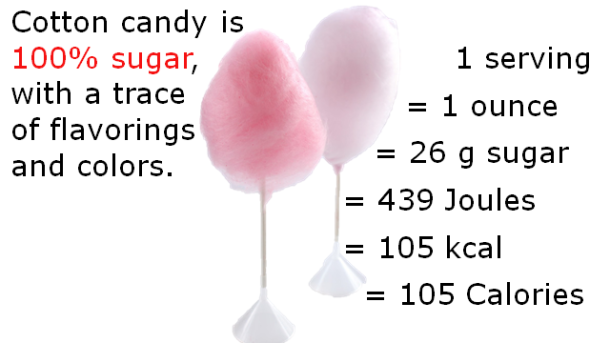A charcuterie board is the perfect accompaniment to any gathering and rivals a cheese plate as a crowd-pleaser. It’s low maintenance, delicious, and will almost certainly have a taste or texture to appeal to the pickiest of palates. Meat comes in an array of textures, fat content, and flavors, which vary species to species and even within the same animal. Flavor profiles of meat can vary wildly and subtleties between different cuts of meat can all be largely explained by chemistry.

Photo credit: willmacdonald18 (Flickr)
What is meat exactly? Meat can loosely mean any type of edible tissue originating from an animal – including everything from chicken feet to cow tongue. The majority of meat we consume, however, is skeletal muscle tissue, comprising roughly 75% water, 20% protein, and 3% fat. While the function of muscle tissue—which is to generate movement—is simple, muscle tissue is a complex system of biochemical machinery.
Muscle tissue consists white and red fibers, which each generate contrasting types of movement. The major differences between the two types of muscle fibers are summarized in the table below.
|
red muscle fibers |
white muscle fibers |
| alternate names |
slow-twitch fibers |
fast-twitch fibers |
| relative size |
thin |
thick |
| movement type |
prolonged, deliberate |
short, high-powered bursts |
| fueled by |
fat + oxygen |
glycogen |
| Requires oxygen? |
absolutely |
yes, but can run anaerobically |
| taste/nutrtion |
fattier, more flavorful |
higher in protein, relatively bland |
| appearance |
dark/red |
white |
The main distinction between these types of tissue is in their function and metabolic demands. Red muscle fibers are designed for endurance—think long distance running—or sustained motion. Red tissue runs on fat and oxygen and absolutely requires oxygen to function properly. Since red muscle tissue demands oxygen, it contains an abundance of a pigmented protein called myoglobin. Myoglobin binds and stores oxygen from the then passes it along to a fat-oxidizing cytochrome that generates ATP to fuel the cell. The more exercise a muscle receives, the higher its demand for oxygen, and the more myoglobin and cytochromes it will contain, leading to a darker appearance.

Photo credit: Harlanh (Flickr) Duck breast – an example of dark meat
White muscle fibers, on the other hand, are specialized for more sporadic and brief energetic demands. These tissues use oxygen to burn glycogen, but can also produce energy anaerobically if needed. However, anaerobic metabolism results in the buildup of lactic acid and limits the endurance of these tissues, which is why they can only be used for short periods. White muscle fibers, unsurprisingly, are what comprise white meat—chicken breast, turkey breast, frog legs, and rabbit meat.

Photo credit: allthingschill (Flickr) Turkey breast
The link between structure, function, and taste can be used to answer the question of why chickens and turkeys have a combination of dark and white meat, but why ducks and geese are all dark meat. They’re all birds, after all, so it’s rather surprising that there’s such a drastic difference in their breast meat, until you consider their habits. Chickens and turkeys are relatively flightless birds. They stand, walk, or run, and use their legs to bear their weight. Their constantly used legs are mainly dark meat and their infrequently used breasts are composed of white meat. Duck and geese, however, are migratory birds whose flight patterns enlist the use of breast muscles to help them stay airborne for extended periods. To aid them in sustained flights, their chests muscles require increased stores of oxygen and myoglobin, making their breast meat dark, in stark contrast to a chicken or turkey’s breast.
The dichotomy of metabolic demands between red and white meat clearly impacts their physiology, but how does this translate into taste? The complexity of the biochemical equipment needed to store fat and oxygen and metabolize fat in red muscle tissue equates to a higher number of enzymes present in the cell that can break down and produce more flavorful compounds when cooked. A piece of dark or red meat is fattier, boasts richer and more complex flavors, and retains moisture far better than white meat.
How else can chemistry explain the different tastes of meat, ranging from the beefy flavors of cow to the gamey flavors of duck breast? It’s a common saying in the culinary world that where there’s fat, there’s flavor. While fat serves as storage for energy, it doubles as storage for flavor. With fat distributed throughout the meat, any fat-soluble flavor or aroma compounds can end up in them providing meat with its unique flavor profile. What winds up in the fat is heavily influenced by diet – cows taste “beefy” as a result of flavor compounds metabolized from grass and forage. Lamb and sheep’s distinctive flavors come from compounds produced by the liver, and the gamey flavors of duck meat are likely derived from intestinal microbes.
Examining meat through a scientific lens allows us to relate some common mantras of biology, chemistry, and cooking: structure and function go hand in hand, fat is where the flavor is, and you are (and you also taste like) what you eat. In the context of meat, physiology and flavor are intertwined and whether you’re a fan of dark or white meat, you can surely appreciate the fascinating connection between animal physiology and flavor.
References Cited:
- McGee, Harold. On food and cooking: the science and lore of the kitchen. New York: Simon & Schuster, 1997. Print.
 About the author: Mai Nguyen is an aspiring food scientist who received her B.S. in biochemistry from the University of Virginia. She hopes to soon escape the bench in pursuit of a more creative and fulfilling career.
About the author: Mai Nguyen is an aspiring food scientist who received her B.S. in biochemistry from the University of Virginia. She hopes to soon escape the bench in pursuit of a more creative and fulfilling career.
Read more by Mai Nguyen